Understanding arterial blood gas (ABG) interpretation begins with mastering one foundational concept: acid-base balance. While ABGs can feel overwhelming at first, every value ultimately ties back to how well the body is maintaining blood pH within a narrow, life-sustaining range. Even small shifts in pH can disrupt cellular function, impair organ systems, and signal serious underlying pathology.
This article breaks down pH balance in a clear, step-by-step way, including what pH means, why it matters, how buffer systems work, and how the lungs and kidneys coordinate compensation.
You will also learn key ABG normal values, the four primary acid-base disorders, how to tell compensation from correction, and a simple framework you can use to interpret ABGs with confidence.
Take our free course to master the basics of ABG interpretation with clear explanations and helpful practice questions.
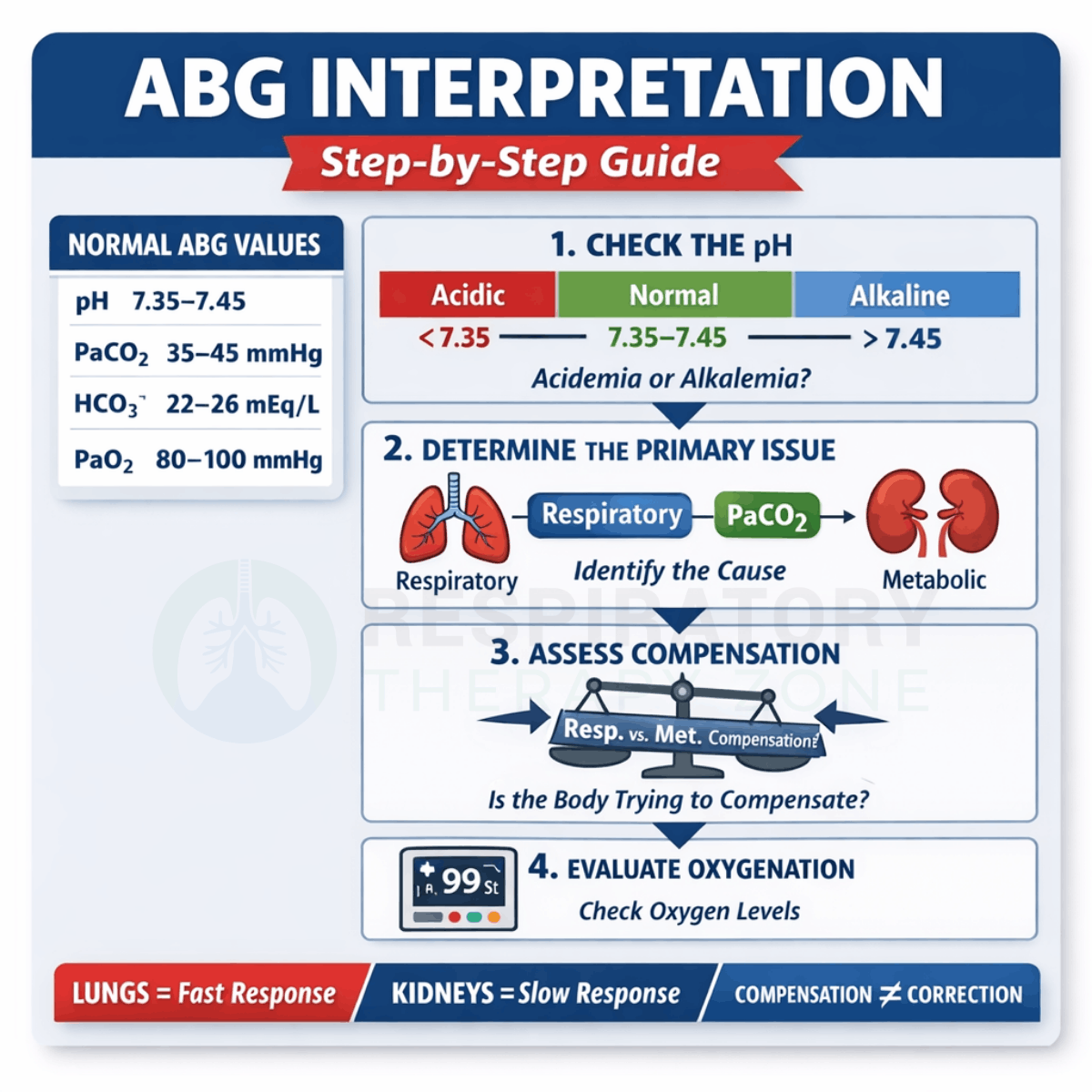
ABG Normal Values (Quick Reference)
Before interpreting any ABG, you need a baseline reference range. These are the core values most commonly used:
- pH: 7.35 to 7.45
- PaCO2: 35 to 45 mmHg
- HCO3-: 22 to 26 mEq/L
- PaO2: 80 to 100 mmHg (varies with age and clinical context)
In ABG interpretation, pH tells you whether the blood is acidemic or alkalemic, PaCO2 reflects the respiratory component, and HCO3- reflects the metabolic component. PaO2 helps you evaluate oxygenation, which is often just as clinically important as the acid-base status.
At the Core of ABG Interpretation: pH Balance
At the core of ABG interpretation is one simple but powerful concept: pH balance. The body is finely tuned to maintain its internal pH within a very narrow range, between 7.35 and 7.45. This balance is essential for normal cellular function. Even small deviations from this range can interfere with enzyme activity, electrolyte balance, and organ performance.
When the pH drops too low (acidemia) or climbs too high (alkalemia), the body experiences physiologic stress. If left uncorrected, these imbalances can become life-threatening.
What Is pH?
The term pH refers to the concentration of hydrogen ions (H+) in the blood. It is a logarithmic scale, which means that for every 0.1 unit change in pH, there is a tenfold change in hydrogen ion concentration.
That is why a change from pH 7.40 to 7.30 is not a minor shift. It represents a significant increase in acidity.
- Low pH means a higher concentration of hydrogen ions, which is acidosis.
- High pH reflects fewer hydrogen ions, which is alkalosis.
Note: The body constantly strives to maintain equilibrium using several mechanisms, including chemical buffers, the lungs, and the kidneys.
Why Acid-Base Balance Matters
Hydrogen ions directly affect the charge and shape of proteins, including enzymes. Since enzymes drive nearly every chemical reaction in the body, a significant pH shift can slow or stop critical physiologic processes. This can interfere with:
- Energy production
- Muscle contraction
- Oxygen delivery
- Nervous system signaling
Clinical effects can be severe:
- Acidosis may cause confusion, lethargy, coma, and impaired cardiac contractility.
- Alkalosis may lead to muscle twitching, paresthesia, cardiac arrhythmias, and seizures.
Note: Maintaining proper pH is not optional. It is central to survival, which is why the body uses powerful, overlapping systems to detect and correct acid-base disturbances.
Buffer Systems of the Body
The body constantly produces acids as byproducts of metabolism. Without protection, the blood would quickly become too acidic to sustain life. To prevent this, the body relies on buffer systems, which minimize changes in pH by neutralizing excess hydrogen ions (H+) or hydroxide ions (OH-).
Several buffer systems contribute to acid-base stability, including:
- Protein buffer system
- Hemoglobin buffer system
- Phosphate buffer system
- Bicarbonate buffer system (the most important in blood)
Bicarbonate Buffer System
The bicarbonate buffer system is the primary buffer system of extracellular fluid and the main one evaluated with an ABG. It maintains equilibrium between carbonic acid (H2CO3) and bicarbonate (HCO3-).
When the blood becomes too acidic (too many H+ ions), bicarbonate binds with hydrogen ions to form carbonic acid. Carbonic acid then converts to water and carbon dioxide, which can be removed by the lungs.
When the blood becomes too alkaline (too few H+ ions), carbonic acid dissociates to release more hydrogen ions, lowering the pH back toward normal.
This system adjusts quickly through the respiratory system and more slowly through the kidneys. In general, buffers act like a first line of defense. They do not fix the underlying cause, but they buy the body time until the lungs and kidneys can make more lasting adjustments.
Primary Acid-Base Disorders
ABG interpretation becomes much easier when you understand the four primary acid-base disorders and what drives them.
Respiratory Acidosis
Respiratory acidosis occurs when PaCO2 rises due to hypoventilation or inadequate CO2 elimination. The primary problem is respiratory, and the kidneys compensate by retaining bicarbonate over time.
Respiratory Alkalosis
Respiratory alkalosis occurs when PaCO2 falls due to hyperventilation. The primary problem is respiratory, and the kidneys compensate by excreting bicarbonate over time.
Metabolic Acidosis
Metabolic acidosis occurs when HCO3- falls due to loss of bicarbonate or accumulation of acid. The primary problem is metabolic, and the lungs compensate quickly by blowing off CO2.
Metabolic Alkalosis
Metabolic alkalosis occurs when HCO3- rises due to excess base or loss of acid. The primary problem is metabolic, and the lungs compensate by retaining CO2 through hypoventilation, within physiologic limits.
Compensation vs. Correction
A common mistake in ABG interpretation is assuming compensation means the problem is fixed. Compensation is the body’s response, not a cure. It is an attempt to stabilize pH while the underlying cause persists.
- Compensation does not eliminate the primary disorder.
- Compensation usually does not return pH completely to normal.
- If pH is within normal range but PaCO2 and HCO3- are abnormal, the patient may have a fully compensated disorder.
Note: In other words, you can see abnormal PaCO2 and abnormal HCO3- with a near-normal pH, and the patient can still be very sick. Your job is to identify which abnormality is primary and which is the compensatory response.
The Role of the Lungs (Respiratory Regulation of CO2)
The lungs play a vital role in maintaining acid-base balance by regulating the level of carbon dioxide (CO2) in the blood. Since CO2 is an acid-forming gas, its concentration directly influences pH.
The lungs act rapidly, within seconds to minutes, making them the body’s fastest system for compensating acid-base imbalances.
How the Lungs Compensate
When the blood becomes too acidic (low pH), the brainstem triggers faster and deeper breathing (hyperventilation). This expels more CO2, reduces hydrogen ion concentration, and raises pH back toward normal.
When the blood becomes too alkaline (high pH), breathing slows (hypoventilation), allowing CO2 to accumulate. This increases hydrogen ions and lowers pH.
This control is regulated primarily by chemoreceptors in the brainstem (central) and the carotid and aortic bodies (peripheral). These receptors monitor pH, PaCO2, and PaO2 and adjust ventilation accordingly.
Examples in Clinical Practice
- In metabolic acidosis (such as diabetic ketoacidosis), patients often develop rapid, deep breathing known as Kussmaul respirations to blow off CO2 and raise pH.
- In metabolic alkalosis (such as vomiting or diuretic overuse), ventilation may slow to retain CO2 and lower pH.
- In COPD, patients may chronically retain CO2. Over time, their bodies adapt, and ventilatory drive may shift more toward oxygen levels than CO2.
Limitations of Respiratory Compensation
- It cannot fully normalize pH when a severe metabolic problem persists.
- It depends on intact respiratory muscles and neural control.
- If ventilation is impaired (such as overdose or neuromuscular disease), CO2 can accumulate rapidly and cause life-threatening respiratory acidosis.
Note: When the problem is prolonged, the kidneys must provide longer-term adjustment.
The Role of the Kidneys (Renal Regulation of HCO3-)
While the lungs provide rapid, short-term regulation of acid-base balance through CO2 control, the kidneys offer slower but more powerful long-term compensation. Their role centers on managing bicarbonate (HCO3-) and excreting hydrogen ions (H+).
Unlike the lungs, which work in minutes, the kidneys take hours to days to fully respond. When they do, their effects are sustained and essential, especially in chronic conditions.
1. Bicarbonate Reabsorption
Each day, the kidneys filter large amounts of bicarbonate into the urine. Under normal conditions, nearly 100% is reabsorbed back into the blood, mostly in the proximal tubules. This preserves the body’s buffering capacity.
- In acidosis, the kidneys increase bicarbonate reabsorption to preserve base and raise pH.
- In alkalosis, the kidneys reduce reabsorption and may allow bicarbonate to be lost in urine to help lower pH.
2. Hydrogen Ion Secretion and New Bicarbonate Production
The kidneys also secrete hydrogen ions into the urine, mainly in the distal tubules and collecting ducts. These hydrogen ions are buffered in urine by phosphate or ammonia.
For every hydrogen ion excreted, a new bicarbonate ion is generated and returned to the blood. This allows the kidneys to create new base, which is especially important in chronic acidosis and ongoing CO2 retention.
Examples in Clinical Practice
- In chronic respiratory acidosis (such as COPD), the kidneys retain bicarbonate to buffer CO2 buildup. A patient may have PaCO2 of 55 mmHg with a pH near 7.37 due to elevated HCO3-.
- In renal failure, the kidneys may be unable to excrete enough H+ or regenerate bicarbonate, worsening metabolic acidosis over time.
- In metabolic alkalosis, the kidneys attempt to excrete bicarbonate and retain H+, but this may be limited by volume depletion or hypokalemia.
Limitations of Renal Compensation
- It is slow, so it is not effective for acute disturbances.
- It depends on adequate kidney function.
- It can be influenced or overridden by diuretics and hormone states (such as aldosterone excess).
A Simple Step-by-Step ABG Interpretation Method
This basic framework turns acid-base physiology into a practical process you can use on exams and in clinical practice.
- Check the pH. Determine whether the patient is acidemic (below 7.35) or alkalemic (above 7.45).
- Identify the primary problem. Compare PaCO2 and HCO3- to the direction of the pH. The PaCO2 moves opposite to the pH (respiratory), and the HCO3- moves with the pH (metabolic).
- Assess compensation. Determine whether the other value is moving in the expected direction to compensate. Remember, compensation is an attempt to normalize pH, not a cure.
- Evaluate oxygenation. Review PaO2 (and SaO2 if provided) to assess oxygenation status and severity.
Common Pitfalls and Clinical Pearls
- Do not ignore oxygenation. A patient can have a mild acid-base disturbance and severe hypoxemia.
- pH is your starting point, not your final answer. Always identify the primary disorder and compensation.
- Lungs are fast, kidneys are slow. Acute changes are often respiratory, chronic patterns often show renal involvement.
- Compensation has limits. If compensation seems excessive or mismatched, consider a mixed disorder.
- Trend matters. One ABG is a snapshot. Serial ABGs reveal progression, response to therapy, and worsening fatigue.
Final Thoughts
Acid-base balance is the cornerstone of ABG interpretation, and understanding how the body regulates pH brings clarity to what can otherwise feel like a complex topic. Buffer systems provide immediate protection, the lungs deliver rapid short-term compensation by controlling CO2, and the kidneys create powerful long-term stability through bicarbonate management and hydrogen ion excretion.
When you combine this physiology with a consistent step-by-step interpretation method, ABGs become far more logical and predictable.
Mastering these principles strengthens exam performance and improves clinical decision-making, allowing you to recognize acid-base disturbances faster, interpret compensation correctly, and respond with greater confidence and precision.
Written by:
John Landry is a registered respiratory therapist from Memphis, TN, and has a bachelor's degree in kinesiology. He enjoys using evidence-based research to help others breathe easier and live a healthier life.
References
- Hopkins E, Sanvictores T, Sharma S. Physiology, Acid Base Balance. [Updated 2022 Sep 12]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025.



