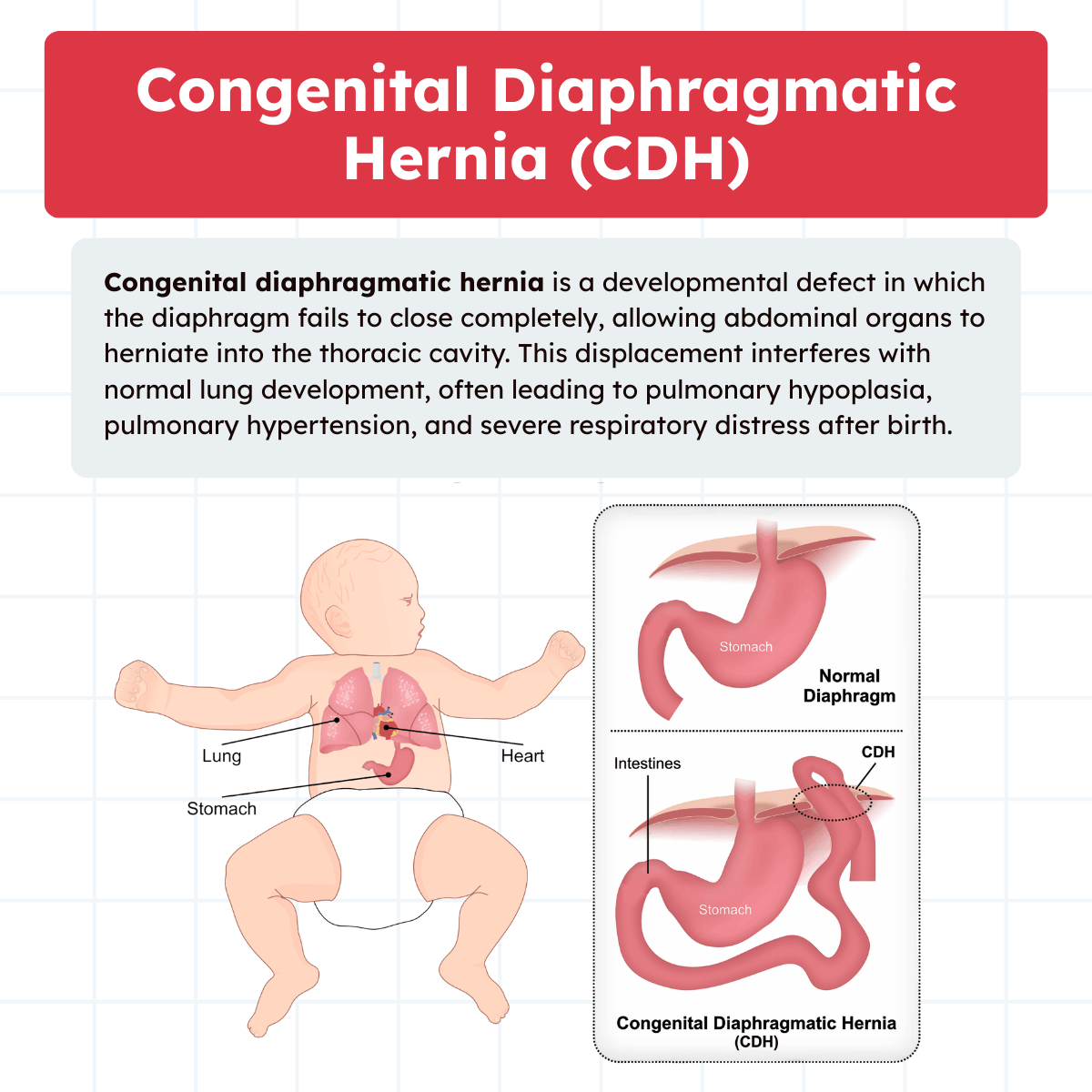
Congenital diaphragmatic hernia (CDH) is a life-threatening developmental disorder that presents most commonly in the neonatal period with severe respiratory distress. Although it is considered a surgical condition, its clinical course is driven largely by pulmonary hypoplasia and pulmonary hypertension, making it highly relevant to respiratory therapists.
From delivery room stabilization to advanced ventilatory strategies and extracorporeal support, respiratory care plays a central role in survival. Understanding the pathophysiology, assessment findings, and management priorities of CDH is essential for anyone involved in neonatal and critical care practice.
What is a Congenital Diaphragmatic Hernia?
A congenital diaphragmatic hernia is a structural defect that occurs when the diaphragm fails to close completely during fetal development. This incomplete formation allows abdominal organs, such as the intestines, stomach, spleen, and sometimes the liver, to herniate into the thoracic cavity.
The presence of abdominal contents in the chest interferes with normal lung development. As a result, infants are born not only with a diaphragmatic defect but also with underdeveloped lungs. This combination of abnormal anatomy and impaired pulmonary growth creates a cascade of respiratory and cardiovascular complications immediately after birth.
CDH is more than simply “a hole in the diaphragm.” It is often associated with profound pulmonary hypoplasia, abnormal pulmonary vasculature, and persistent pulmonary hypertension of the newborn. These factors largely determine the severity of illness and overall prognosis.
Types of Congenital Diaphragmatic Hernia
Bochdalek Hernia
A Bochdalek hernia is the most common form. It occurs in the posterolateral portion of the diaphragm and is usually found on the left side. Because the liver is on the right side of the abdomen, left-sided defects often allow more abdominal contents to enter the thoracic cavity, contributing to more severe lung compression.
Morgagni Hernia
A Morgagni hernia is less common and occurs in the anterior and medial portion of the diaphragm. It may occur on either side and is sometimes less severe. In rare cases, infants may even be asymptomatic.
Right-sided hernias may appear less severe in some cases because the liver can partially block the defect, limiting the volume of abdominal contents that enter the thorax. However, right-sided CDH can still be associated with significant pulmonary hypoplasia and pulmonary hypertension.
Pathophysiology
The primary issue in congenital diaphragmatic hernia is not just the defect in the diaphragm, but the abnormal development of the lungs. During fetal life, abdominal organs herniate into the thoracic cavity, compressing the developing lungs. This mechanical compression leads to pulmonary hypoplasia, characterized by a reduced number of alveoli and a diminished pulmonary vascular bed.
In addition to smaller lungs, the pulmonary arteries are structurally abnormal and prone to vasoconstriction. After birth, this results in elevated pulmonary vascular resistance and persistent pulmonary hypertension. The high pressures promote right-to-left shunting through fetal circulatory pathways, worsening hypoxemia.
Because of these combined factors, oxygenation is severely impaired. Even with adequate ventilation, the limited lung tissue and elevated pulmonary pressures prevent effective gas exchange. Therefore, respiratory failure in CDH is primarily driven by underdeveloped lungs and abnormal pulmonary circulation rather than the diaphragmatic defect alone.
Clinical Presentation
Most cases of CDH are diagnosed prenatally using ultrasonography. However, some infants present at birth with acute respiratory distress.
Common physical findings include:
- Severe tachypnea
- Retractions and nasal flaring
- Grunting
- Cyanosis
- Scaphoid (sunken) abdomen due to abdominal contents in the thorax
- Decreased or absent breath sounds on the affected side
- Displaced heart sounds
- Mediastinal shift
Note: A chest radiograph confirms the diagnosis. Imaging typically shows bowel loops within the thoracic cavity, mediastinal displacement, and varying degrees of lung collapse or atelectasis. Arterial blood gas results often reveal respiratory acidosis with hypoxemia.
Why CDH Is Relevant to Respiratory Therapists
Although CDH requires surgical repair, respiratory management determines survival in the early stages. Respiratory therapists are central to stabilization, ventilator management, oxygenation strategies, and coordination of advanced therapies.
Delivery Room Management
Immediate priorities include:
- Endotracheal intubation
- Avoidance of bag-valve-mask ventilation
- Placement of an orogastric tube connected to continuous suction
Note: Positive pressure ventilation via mask is avoided because it can insufflate the stomach and intestines, worsening lung compression. Early intubation prevents further compromise and allows controlled ventilation.
Mechanical Ventilation
Conventional ventilation is initiated using lung-protective strategies. The goal is to minimize barotrauma and volutrauma while maintaining adequate oxygenation and ventilation. Permissive hypercapnia may be accepted to reduce ventilator-induced lung injury. Careful monitoring of peak inspiratory pressures, tidal volumes, and oxygen concentration is essential.
In more severe cases, high-frequency ventilation (HFV), particularly high-frequency oscillatory ventilation (HFOV), may be used. HFV provides adequate gas exchange with lower tidal volumes, reducing the risk of further lung injury.
Pulmonary Hypertension Management
Pulmonary hypertension is a major determinant of outcome in CDH. Elevated PVR leads to right-to-left shunting and refractory hypoxemia.
Management may include:
- Optimization of oxygenation
- Careful ventilation strategies
- Sedation and paralysis
- Inhaled nitric oxide in select cases
Note: Despite these interventions, some infants continue to deteriorate.
Extracorporeal Membrane Oxygenation (ECMO)
In severe cases, extracorporeal membrane oxygenation may be necessary. ECMO provides cardiopulmonary support by oxygenating blood outside the body, allowing the lungs to rest and pulmonary pressures to decrease.
Some centers perform surgical repair while the infant is on ECMO. Others prefer to wean the infant off ECMO before surgery. Current trends increasingly favor stabilization and weaning from ECMO prior to operative repair when possible.
Respiratory therapists play an important role in ventilator adjustments during ECMO support and in the transition back to conventional ventilation.
Surgical Repair
Surgical correction is typically delayed for several days to allow pulmonary vascular resistance to decrease. During surgery, the diaphragmatic defect is closed. In large defects, a patch may be required.
The timing of surgery depends on stabilization of oxygenation and pulmonary pressures. Infants with severe pulmonary hypertension may not tolerate immediate repair.
Long-Term Complications
Survivors of CDH may experience ongoing complications, including:
- Chronic lung disease
- Recurrent pulmonary infections
- Severe gastroesophageal reflux
- Feeding difficulties
- Growth delays
- Scoliosis
Note: The association with scoliosis suggests that CDH may represent a broader developmental defect rather than an isolated diaphragmatic issue. Long-term follow-up often requires a multidisciplinary team, including pulmonologists, surgeons, and respiratory therapists.
Prognosis and Survival Factors
Survival depends on several complex variables, including:
- Degree of pulmonary hypoplasia
- Presence of liver herniation into the thorax
- Fetal head-to-lung ratio
- Initial oxygenation and carbon dioxide levels
- Severity of pulmonary hypertension
Note: Despite advancements in ventilation strategies and ECMO technology, mortality remains significant. Early recognition, coordinated care, and careful ventilator management improve outcomes.
The Role of the Respiratory Therapist in Ongoing Care
Respiratory therapists are involved in:
- Neonatal resuscitation
- Mechanical ventilation management
- Monitoring oxygenation and ventilation
- Implementing lung-protective strategies
- Assisting with HFV
- Supporting ECMO care
- Educating families
In neonatal intensive care units, respiratory therapists must understand the delicate balance between oxygenation and lung protection. Excessive ventilation pressures can worsen lung injury, while inadequate support can exacerbate hypoxemia and acidosis.
The ability to interpret blood gases, recognize worsening pulmonary hypertension, and collaborate with neonatologists is critical.
Congenital Diaphragmatic Hernia Practice Questions
1. What is congenital diaphragmatic hernia (CDH)?
Congenital diaphragmatic hernia is a birth defect in which incomplete formation of the diaphragm allows abdominal organs to herniate into the thoracic cavity.
2. Why does CDH commonly cause severe respiratory distress in newborns?
Herniation of abdominal contents into the chest compresses the lungs, leading to pulmonary hypoplasia and impaired gas exchange.
3. What are the two primary types of congenital diaphragmatic hernia?
The two main types are Bochdalek hernia and Morgagni hernia.
4. What characterizes a Bochdalek hernia?
A Bochdalek hernia is a posterolateral defect, most commonly occurring on the left side.
5. What characterizes a Morgagni hernia?
A Morgagni hernia is an anterior and medial defect that may occur on either side.
6. Why are right-sided diaphragmatic hernias sometimes less severe?
The liver may partially block the defect, limiting herniation of abdominal contents into the thorax.
7. What is pulmonary hypoplasia in CDH?
Pulmonary hypoplasia refers to underdeveloped lungs with reduced alveoli and pulmonary vasculature.
8. How does pulmonary hypoplasia contribute to hypoxemia?
Fewer alveoli and vessels impair oxygen exchange and increase pulmonary vascular resistance.
9. Why is pulmonary hypertension common in CDH?
Abnormal pulmonary vascular development and hypoxia increase pulmonary vascular resistance.
10. What prenatal test can detect most cases of CDH?
Prenatal ultrasonography can often diagnose CDH in utero.
11. What is a scaphoid abdomen in CDH?
A scaphoid abdomen appears sunken because abdominal organs are displaced into the thoracic cavity.
12. Why are breath sounds decreased or absent on the affected side?
The herniated organs compress the lung, reducing ventilation.
13. Why may heart sounds be displaced in CDH?
Mediastinal shift occurs as abdominal contents push the heart away from the defect.
14. What radiographic finding confirms CDH?
Chest x-ray typically shows bowel loops within the thoracic cavity and mediastinal shift.
15. What arterial blood gas finding is common in severe CDH?
Respiratory acidosis with hypoxemia is commonly observed.
16. Why should bag-valve-mask ventilation be avoided in suspected CDH?
Positive-pressure mask ventilation can insufflate the stomach and worsen lung compression.
17. What is the first airway management step in CDH?
Immediate endotracheal intubation is recommended.
18. Why is an orogastric tube inserted in CDH management?
Continuous suction decompresses the stomach and reduces thoracic compression.
19. What ventilatory strategy may be beneficial in CDH?
High-frequency oscillatory ventilation may improve oxygenation while minimizing lung injury.
20. When is extracorporeal membrane oxygenation (ECMO) considered?
ECMO is used in severe cases with refractory hypoxemia or pulmonary hypertension.
21. Why is surgical repair often delayed in CDH?
Stabilization and reduction of pulmonary hypertension are prioritized before surgery.
22. At what general time frame is surgical repair commonly performed in stable infants?
Repair is often performed several days after birth once cardiopulmonary status stabilizes.
23. What complication may survivors of CDH develop later in life?
Survivors may develop scoliosis or severe gastroesophageal reflux.
24. What is the fetal head-to-lung ratio used for in CDH?
It helps estimate the severity of pulmonary hypoplasia prenatally.
25. Why does CDH increase pulmonary vascular resistance (PVR)?
Abnormal lung development and hypoxia lead to persistent pulmonary hypertension.
26. What are common signs of respiratory distress in CDH?
Tachypnea, retractions, nasal flaring, grunting, and cyanosis are common.
27. Why may mediastinal shift be life-threatening in CDH?
Shifted structures impair both ventilation and cardiac output.
28. What hemodynamic instability may occur in severe CDH?
Persistent pulmonary hypertension can cause right-to-left shunting and severe hypoxemia.
29. Why is CDH considered a neonatal emergency?
Rapid respiratory compromise can occur shortly after birth.
30. What is the overall prognosis of CDH?
Prognosis varies but mortality remains significant, particularly in severe pulmonary hypoplasia cases.
31. What embryologic defect leads to congenital diaphragmatic hernia (CDH)?
Failure of complete fusion of the pleuroperitoneal membranes during fetal development results in a diaphragmatic defect.
32. Which side is most commonly affected in CDH?
The left side is most commonly affected due to earlier closure of the right pleuroperitoneal canal.
33. How does herniation of the liver into the thorax affect prognosis?
Liver herniation is associated with more severe pulmonary hypoplasia and a poorer prognosis.
34. What cardiovascular complication frequently accompanies CDH?
Persistent pulmonary hypertension of the newborn (PPHN) commonly occurs.
35. Why is gentle ventilation emphasized in CDH management?
Overdistention of hypoplastic lungs can worsen barotrauma and increase pulmonary vascular resistance.
36. What is permissive hypercapnia in the context of CDH?
Allowing mildly elevated PaCO₂ levels to minimize ventilator-induced lung injury.
37. Why is high peak inspiratory pressure avoided in CDH?
Excessive pressures can cause pneumothorax and further compromise fragile lungs.
38. What oxygen saturation targets are typically maintained in CDH?
Preductal oxygen saturation is often targeted between 85% and 95% to balance oxygenation and avoid hyperoxia.
39. Why is preductal and postductal SpO₂ monitoring important in CDH?
It helps detect right-to-left shunting through the ductus arteriosus.
40. What echocardiographic finding may be seen in CDH with pulmonary hypertension?
Elevated right ventricular pressures and tricuspid regurgitation may be present.
41. How does pulmonary hypoplasia affect lung compliance in CDH?
The lungs are stiff and poorly compliant due to underdevelopment.
42. What is the purpose of sedation and neuromuscular blockade in severe CDH?
They reduce oxygen consumption and prevent ventilator asynchrony.
43. Why is inhaled nitric oxide sometimes used in CDH?
It may reduce pulmonary hypertension by selectively dilating pulmonary vessels.
44. What complication may result from aggressive ventilation in CDH?
Pneumothorax is a common complication.
45. Why is immediate surgical repair not performed in unstable infants?
Stabilization of respiratory and hemodynamic status improves surgical outcomes.
46. What does mediastinal shift indicate on chest imaging?
Significant herniation of abdominal contents into the thoracic cavity.
47. How does CDH affect alveolar-capillary surface area?
It significantly reduces surface area for gas exchange.
48. Why is careful fluid management important in CDH?
Fluid overload can worsen pulmonary edema and oxygenation.
49. What is the role of surfactant in CDH management?
Surfactant may be considered if respiratory distress syndrome coexists.
50. How does CDH influence long-term pulmonary function?
Survivors may have chronic lung disease and reduced exercise tolerance.
51. Why are serial arterial blood gases important in CDH?
They guide ventilation and oxygenation adjustments.
52. What metabolic abnormality may occur due to hypoxemia in CDH?
Metabolic acidosis may develop secondary to poor tissue perfusion.
53. How does CDH impact cardiac output?
Severe pulmonary hypertension can impair right ventricular output and reduce systemic perfusion.
54. What imaging modality may supplement chest radiography in CDH?
Echocardiography assesses cardiac function and pulmonary pressures.
55. Why is minimal handling recommended in severe CDH?
Stimulation can increase pulmonary vascular resistance and worsen hypoxemia.
56. What long-term gastrointestinal complication is common in CDH survivors?
Gastroesophageal reflux disease is frequently observed.
57. How does CDH affect ventilation-perfusion matching?
Significant V/Q mismatch occurs due to hypoplastic and compressed lung tissue.
58. What prenatal intervention has been explored for severe CDH?
Fetal endoscopic tracheal occlusion has been studied to stimulate lung growth.
59. Why is early diagnosis of CDH critical?
Preparation for immediate neonatal resuscitation improves outcomes.
60. What structural abnormality of the diaphragm defines CDH?
A persistent defect allowing communication between abdominal and thoracic cavities.
61. Why may bowel sounds be heard in the chest of an infant with CDH?
Herniated intestines occupy the thoracic cavity.
62. How does CDH influence systemic oxygen delivery?
Reduced lung development and pulmonary hypertension impair oxygenation.
63. Why is capillary refill time monitored in severe CDH?
It helps assess systemic perfusion and cardiac output.
64. What is a common cause of mortality in CDH?
Refractory pulmonary hypertension and respiratory failure.
65. Why must clinicians avoid excessive oxygen exposure in CDH?
Hyperoxia may worsen oxidative injury and pulmonary vascular reactivity.
66. What percentage of congenital diaphragmatic hernias are classified as Bochdalek defects?
Approximately 80–85% of cases are Bochdalek hernias.
67. Why is bag-mask ventilation contraindicated in suspected CDH?
It can insufflate air into the stomach and intestines, worsening lung compression.
68. What immediate step should be taken when CDH is suspected at birth?
Prompt endotracheal intubation with avoidance of positive-pressure mask ventilation.
69. How does pulmonary vascular remodeling contribute to CDH pathophysiology?
Thickened pulmonary arterioles increase pulmonary vascular resistance and worsen pulmonary hypertension.
70. Why is systemic hypotension concerning in infants with CDH?
It may indicate right ventricular failure and poor systemic perfusion.
71. What role does prostaglandin E1 play in certain CDH cases?
It may be used to maintain ductal patency in severe pulmonary hypertension.
72. Why is thermoregulation important in infants with CDH?
Hypothermia increases oxygen consumption and pulmonary vasoconstriction.
73. What blood gas pattern is commonly seen in severe CDH?
Respiratory acidosis with hypoxemia.
74. How does increased pulmonary vascular resistance affect shunting in CDH?
It promotes right-to-left shunting across the ductus arteriosus and foramen ovale.
75. Why is careful ventilator monitoring essential in CDH management?
To prevent volutrauma and barotrauma in hypoplastic lungs.
76. What complication may arise from prolonged ECMO use in CDH?
Bleeding due to systemic anticoagulation.
77. Why are inotropic agents sometimes required in CDH?
To support cardiac output in the presence of right ventricular dysfunction.
78. How does CDH affect thoracic cavity development?
Herniated abdominal organs impair normal lung growth.
79. What prenatal measurement helps predict CDH severity?
The observed-to-expected lung-to-head ratio (O/E LHR).
80. Why is enteral feeding often delayed in severe CDH?
Respiratory instability and gastrointestinal dysfunction may be present.
81. What postoperative complication is common after CDH repair?
Recurrence of the hernia.
82. How does chronic pulmonary hypertension impact long-term outcomes in CDH survivors?
It may lead to persistent exercise intolerance and right heart strain.
83. Why is sedation minimized when possible in recovering CDH infants?
To facilitate spontaneous breathing and neurological assessment.
84. What imaging finding suggests right ventricular strain in CDH?
Right ventricular enlargement on echocardiography.
85. Why is nitric oxide therapy not universally effective in CDH?
Structural pulmonary hypoplasia may limit its response.
86. How does CDH influence compliance of the chest wall?
The affected side may demonstrate decreased expansion.
87. What is the purpose of arterial line placement in severe CDH?
Continuous blood pressure monitoring and frequent blood gas sampling.
88. Why is metabolic acidosis dangerous in CDH?
It increases pulmonary vascular resistance and worsens hypoxemia.
89. What long-term musculoskeletal complication may occur in CDH survivors?
Scoliosis, often toward the side of the defect.
90. Why is careful monitoring of urine output important in CDH?
It reflects renal perfusion and overall hemodynamic status.
91. How does diaphragmatic repair affect pulmonary mechanics?
It restores separation of thoracic and abdominal cavities, allowing lung expansion.
92. Why are neonates with CDH at risk for feeding difficulties?
Gastroesophageal reflux and poor coordination are common.
93. What is the impact of right-sided CDH compared to left-sided CDH?
Right-sided defects may be harder to detect prenatally but can still be severe.
94. Why is gradual weaning of ventilatory support recommended postoperatively?
To prevent sudden increases in pulmonary vascular resistance.
95. What factor most strongly influences survival in CDH?
The degree of pulmonary hypoplasia and pulmonary hypertension.
96. Why is central venous access often required in CDH?
For medication administration and hemodynamic monitoring.
97. How does CDH affect surfactant production?
Hypoplastic lungs may have reduced surfactant availability.
98. Why are serial echocardiograms performed in CDH patients?
To monitor pulmonary pressures and right ventricular function.
99. What is the role of permissive hypoxemia in CDH management?
To avoid excessive ventilator pressures while maintaining adequate tissue oxygenation.
100. What is a key respiratory therapy goal in CDH?
Optimize oxygenation while minimizing ventilator-induced lung injury.
Final Thoughts
Congenital diaphragmatic hernia is a serious neonatal condition in which the primary threat is not the diaphragmatic defect itself, but the profound pulmonary hypoplasia and pulmonary hypertension that accompany it. These infants face immediate respiratory compromise due to underdeveloped lungs and elevated pulmonary vascular resistance.
For respiratory therapists, CDH demands careful assessment, lung-protective ventilation, and close monitoring of oxygenation and acid-base status. Outcomes improve when respiratory care is precise, coordinated, and evidence-based. A thorough understanding of the disease process allows therapists to anticipate complications and contribute meaningfully to stabilization, recovery, and long-term respiratory health.
Written by:
John Landry is a registered respiratory therapist from Memphis, TN, and has a bachelor's degree in kinesiology. He enjoys using evidence-based research to help others breathe easier and live a healthier life.
References
- Dumpa V, Chandrasekharan P. Congenital Diaphragmatic Hernia. [Updated 2023 Aug 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025.

