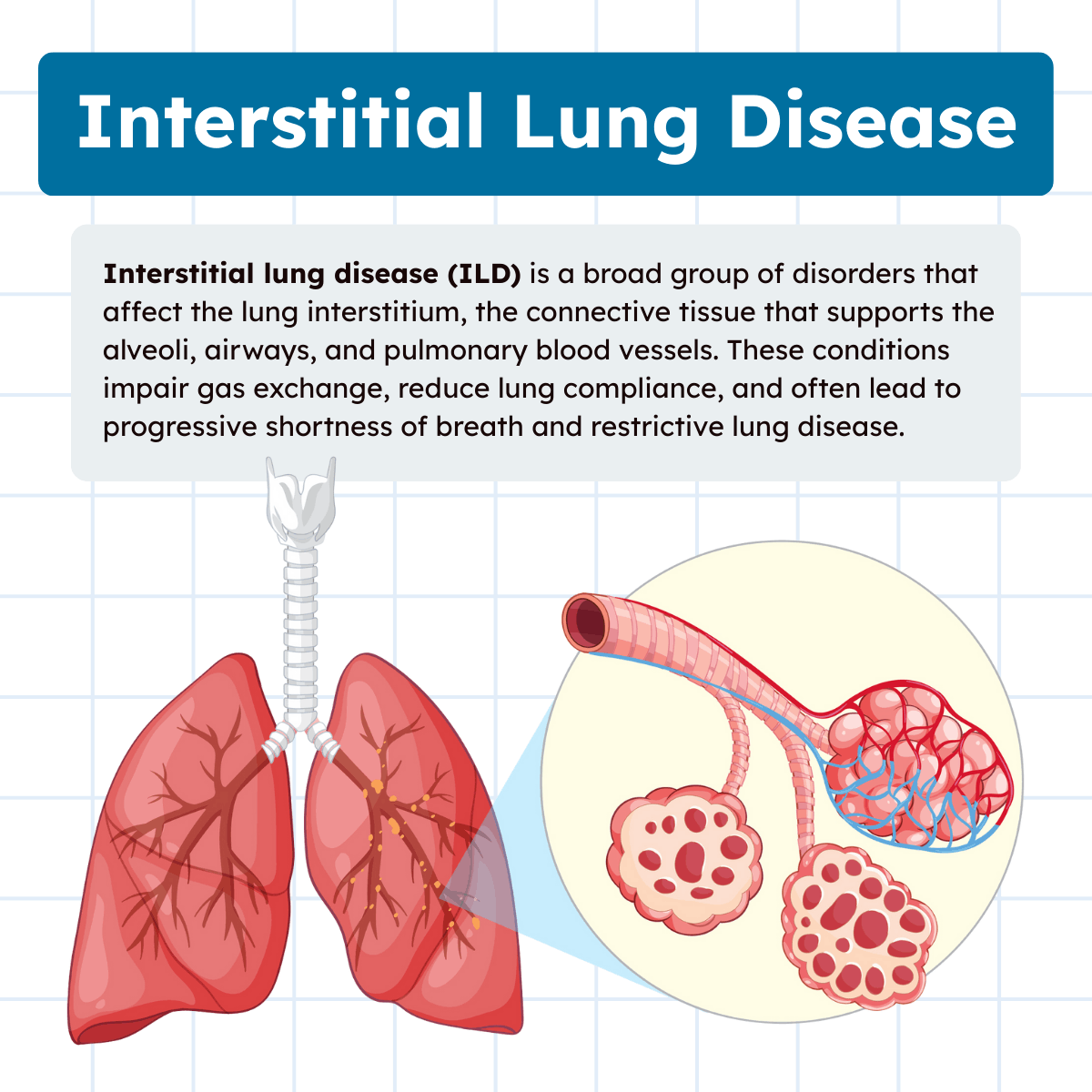
Interstitial lung disease (ILD) refers to a broad group of disorders that primarily affect the interstitium of the lungs—the delicate framework that supports the alveoli, bronchioles, and pulmonary vessels. Although these diseases differ in cause and progression, they share common structural and functional consequences that significantly impair gas exchange.
For respiratory therapists, ILD represents a critical intersection of radiographic interpretation, pulmonary mechanics, oxygen therapy, and long-term patient management.
Understanding ILD is essential not only for recognizing characteristic clinical and imaging findings, but also for optimizing respiratory care, monitoring disease progression, and supporting patients through chronic and often progressive lung disease.
What Is the Interstitium?
To understand interstitial lung disease, it is first important to understand the structure it affects. The interstitium is the connective tissue framework of the lung. It surrounds and supports the alveoli, pulmonary capillaries, bronchioles, and lymphatic structures. This network is extremely thin under normal conditions, allowing oxygen and carbon dioxide to diffuse efficiently between the alveoli and the bloodstream.
The smallest functional unit of the lung is the secondary pulmonary lobule. Each lobule contains alveoli and alveolar ducts arranged around a central bronchiole and pulmonary arteriole. These structures are bordered by intralobular septa, thin sheets of connective tissue that are normally invisible on a standard chest x-ray. When disease or fluid affects these septa, they become radiographically apparent and serve as key diagnostic clues.
What is Interstitial Lung Disease?
Interstitial lung disease is not a single diagnosis but an umbrella term for a large group of disorders that involve inflammation and/or fibrosis of the lung interstitium. These conditions primarily affect the lower respiratory tract and interfere with normal lung expansion and gas exchange.
Unlike alveolar diseases, which fill the airspaces with fluid, pus, or blood, ILD thickens or scars the supporting tissue of the lung. Over time, this process stiffens the lungs, reduces compliance, and increases the work of breathing. Many forms of ILD are chronic and progressive, leading to long-term respiratory impairment and, in advanced cases, respiratory failure.
Radiographic Features of Interstitial Lung Disease
Chest imaging plays a central role in the evaluation of ILD. Although chest x-rays are often the first imaging modality used, they rarely establish a specific diagnosis. Instead, they provide patterns that raise suspicion for interstitial involvement.
Common chest x-ray patterns seen in ILD include:
- Reticular pattern: A network of fine lines representing thickened interstitial structures
- Nodular pattern: Small, poorly defined nodules scattered throughout the lung fields
- Reticulonodular pattern: A combination of lines and nodules
- Honeycombing: Cystic air spaces with well-defined walls, typically seen at the lung periphery
Honeycombing is particularly important because it represents irreversible fibrosis and is considered a hallmark of end-stage interstitial lung disease. Another key radiographic finding is reduced lung volumes, which helps distinguish ILD from obstructive lung diseases such as COPD.
Kerley Lines and Interstitial Edema
Interstitial involvement is not limited to chronic fibrotic diseases. In conditions such as pulmonary edema caused by left-sided heart failure, fluid can accumulate within the interlobular septa. This results in the appearance of Kerley B lines, which are short, horizontal lines seen near the lung bases and perpendicular to the pleura.
While pulmonary edema is not classified as ILD, understanding these interstitial changes is important for respiratory therapists. It reinforces how alterations in the interstitium can dramatically affect lung mechanics and oxygenation, even when the alveoli themselves are not filled with fluid.
High-Resolution CT and ILD
High-resolution computed tomography (HRCT) has become the most important imaging tool in the evaluation of suspected interstitial lung disease. HRCT provides detailed visualization of lung parenchyma and can identify patterns that are not visible on standard chest x-rays.
HRCT is especially valuable in diagnosing idiopathic pulmonary fibrosis (IPF). Characteristic findings include:
- Predominant involvement of the lower lobes
- Subpleural distribution of abnormalities
- Presence of honeycombing
- Minimal ground-glass opacity in advanced disease
Note: For respiratory therapists, HRCT findings often explain clinical symptoms such as progressive dyspnea, reduced lung compliance, and refractory hypoxemia.
Common Causes of Interstitial Lung Disease
Interstitial lung diseases arise from a wide range of causes, and many remain idiopathic, meaning no clear cause is identified. Major categories include:
Occupational and environmental exposures
Long-term inhalation of harmful particles can trigger interstitial inflammation and fibrosis. Examples include asbestos exposure (asbestosis) and silica exposure (silicosis). These conditions often develop years after exposure and are progressive.
Connective tissue and autoimmune diseases
Systemic conditions such as rheumatoid arthritis and scleroderma can involve the lungs and lead to interstitial changes. In these cases, ILD may be one of several organ systems affected.
Idiopathic interstitial pneumonias
Sarcoidosis and idiopathic pulmonary fibrosis are among the most common forms of ILD with no known cause. These diseases often present insidiously and may be advanced by the time they are diagnosed.
Pulmonary Function Changes in ILD
Pulmonary function testing provides critical information about the physiologic impact of interstitial lung disease. ILD typically produces a restrictive pattern, characterized by:
- Decreased total lung capacity (TLC)
- Reduced vital capacity (VC)
- Normal or increased FEV1/FVC ratio
Diffusion capacity for carbon monoxide (DLCO) is often significantly reduced due to thickening or destruction of the alveolar-capillary membrane. This reduction helps explain why many ILD patients experience severe exertional dyspnea even when resting oxygen saturation appears relatively normal.
Clinical Presentation and Symptoms
Patients with interstitial lung disease often present with nonspecific symptoms, which can delay diagnosis. Common clinical features include:
- Progressive shortness of breath, especially with exertion
- Persistent dry cough
- Fatigue and exercise intolerance
- Fine inspiratory crackles on auscultation
In advanced disease, patients may develop hypoxemia at rest, digital clubbing, pulmonary hypertension, and signs of right heart strain. These manifestations significantly affect quality of life and require ongoing respiratory management.
Types of Interstitial Lung Disease
Interstitial lung disease includes more than 200 distinct conditions, but they are often grouped into categories based on cause, pathophysiology, and clinical behavior.
Understanding these categories helps respiratory therapists anticipate disease progression, interpret imaging and pulmonary function findings, and tailor patient care more effectively.
Idiopathic Interstitial Pneumonias (IIPs)
These are interstitial lung diseases with no identifiable cause. The most well-known is idiopathic pulmonary fibrosis (IPF), a chronic, progressive fibrotic disease characterized by worsening dyspnea, reduced lung volumes, and honeycombing on imaging.
Other IIPs include nonspecific interstitial pneumonia (NSIP) and cryptogenic organizing pneumonia (COP). These conditions vary in severity and response to therapy, with fibrotic forms generally carrying a poorer prognosis.
Occupational and Environmental Lung Diseases
Long-term exposure to inhaled particles can lead to interstitial inflammation and fibrosis. Examples include asbestosis from asbestos exposure and silicosis from inhalation of silica dust. These diseases often develop years after exposure and are typically progressive, making early recognition and exposure history especially important.
Connective Tissue Disease–Associated ILD
Autoimmune and connective tissue disorders can involve the lungs and produce interstitial changes. Rheumatoid arthritis, scleroderma, systemic lupus erythematosus, and polymyositis are common examples. In some patients, pulmonary involvement may be the first or most severe manifestation of systemic disease.
Granulomatous Interstitial Lung Diseases
Sarcoidosis is the most common condition in this category. It is characterized by noncaseating granulomas and often presents with bilateral hilar lymphadenopathy, upper-lobe involvement, and restrictive lung physiology. Disease severity ranges from self-limiting to chronic, progressive lung damage.
Hypersensitivity Pneumonitis
This form of ILD results from repeated inhalation of organic antigens such as mold, bird proteins, or agricultural dusts. Acute, subacute, and chronic forms exist. Chronic hypersensitivity pneumonitis can lead to fibrosis and permanent lung damage if exposure continues.
Drug- and Radiation-Induced ILD
Certain medications, including some chemotherapeutic agents and antiarrhythmics, can cause interstitial lung injury. Radiation therapy to the chest may also lead to radiation pneumonitis and subsequent fibrosis.
Note: While these categories help organize ILD, overlap is common. For respiratory therapists, recognizing patterns rather than memorizing individual diseases is often the most practical approach to understanding and managing interstitial lung disease.
Relevance to Respiratory Therapists
Interstitial lung disease is highly relevant to respiratory therapists because it affects nearly every aspect of respiratory care. Therapists are often involved early in the diagnostic process through pulmonary function testing and oxygen assessment.
Oxygen therapy plays a central role in managing ILD, particularly during exertion and sleep. Many patients require ambulatory oxygen long before they need continuous oxygen support. Respiratory therapists are essential in titrating oxygen delivery, educating patients, and monitoring response to therapy.
In acute settings, ILD patients may present with respiratory failure or acute exacerbations. Mechanical ventilation in this population is especially challenging due to poor lung compliance and high risk of ventilator-induced lung injury. Lung-protective strategies, careful monitoring of pressures, and close collaboration with the healthcare team are critical.
Disease Progression and Long-Term Care
Most forms of interstitial lung disease are chronic and progressive. While some inflammatory ILDs may respond to corticosteroids or immunosuppressive therapy, fibrotic diseases often continue to worsen despite treatment.
Respiratory therapists play a vital role in long-term disease management, including pulmonary rehabilitation, breathing retraining, and patient education. Teaching energy conservation techniques and proper use of oxygen equipment can significantly improve daily functioning and independence.
Interstitial Lung Disease Practice Questions
1. What does ILD stand for?
ILD stands for Interstitial Lung Disease. It’s an abbreviation that we’ll use throughout the remainder of this study guide.
2. Interstitial lung diseases are a diverse group of illnesses that can be organized into groups based on what?
They are organized based on related causes.
3. Interstitial lung diseases generally cause what?
They cause chronic, progressive dyspnea on exertion and a nonproductive cough.
4. What breath sounds are common with ILD?
Fine, inspiratory crackles
5. What is the most common chest radiograph finding in ILD?
Diffuse reticular or reticulonodular infiltrates with reduced lung volumes.
6. What are the PFTs like for a patient with ILD?
Pulmonary function testing usually reveals restrictive physiology and decreased diffusion capacity; however, other patterns may be seen.
7. What are the causes of ILD?
The causes most frequently occur from exposure (tobacco, hypersensitivity pneumonitis antigens, silica, asbestos), autoimmune dysfunction (sarcoidosis, connective tissue diseases), and abnormal injury healing (IPF).
8. What is the specific treatment for ILD depend upon?
It depends on the underlying disease and may consist of an immunosuppressive.
9. What is the most common treatment for interstitial lung disease?
A lung transplant
10. What is a common physical finding of ILD?
Bibasilar inspiratory dry crackles in the bases.
11. What is the classification of ILD based on?
Location
12. What is the most effective way to diagnose ILD?
Open lung biopsy
13. How does ILD affect blood pressure?
Hypertension is a symptom of ILD.
14. What form of ILD is typically seen in 50 to 70-year-old men with progressive dyspnea and bibasilar crackles?
Idiopathic Pulmonary Fibrosis
15. What form of ILD presents with intermittent episodes of dyspnea and is seen in people who live on farms?
Hypersensitivity pneumonitis
16. What is the most important thing to find when diagnosing hypersensitivity pneumonitis?
The patient’s history and work history.
17. What does the diagnosis of drug-induced interstitial lung disease rely on?
The exclusion of all other processes.
18. What type of ILD is commonly seen in coal miners, potters, and drill workers?
Silicosis
19. What is the primary goal of ILD treatment?
To prevent irreversible lung damage
20. The term Interstitial Lung Disease (ILD) refers to a broad group of inflammatory lung disorders that are characterized by what?
Acute, subacute, or chronic inflammatory infiltration of the alveolar walls by cells, fluid, and connective tissue.
21. What are some other names for Interstitial Lung Diseases?
Diffuse interstitial lung disease, fibrotic interstitial lung disease, pulmonary fibrosis, or pneumoconiosis.
22. If left untreated, an inflammatory process in the lungs can progress to what?
Irreversible pulmonary fibrosis
23. What are the major pathologic or structural changes of the lungs associated with interstitial lung disease?
Destruction of the alveoli and adjacent pulmonary capillaries; fibrotic thickening of the respiratory bronchioles, alveolar ducts, and alveoli; granulomas, honeycombing, and cavity formation; fibrocalcific pleural plaques (with asbestos); bronchospasm and excessive bronchial secretions.
24. How is the acute stage of ILD characterized?
Edema and the infiltration of a variety of white blood cells in the alveolar walls and interstitial spaces.
25. How is the chronic stage of ILD characterized?
In addition to the inflammation and infiltration of numerous white blood cells, some fibroblasts may be present in the alveolar walls and interstitial spaces.
26. What can occur after the chronic stage of ILD?
Further interstitial thickening, fibrosis, granulomas, and in some cases, honeycombing and cavity formation.
27. The exposure to what organic materials can cause ILD?
Moldy hay, silage, moldy sugar cane, mushroom compost, barley, cheese, wood pulp, bark, dust, cork dust, bird droppings, and paint.
28. The exposure to what medications can cause ILD?
Antibiotics, anti-inflammatory agents, cardiovascular agents, chemotherapy agents, and illicit drugs.
29. What connective tissue diseases can lead to ILD?
Scleroderma, rheumatoid arthritis, Sjogren’s syndrome, systemic lupus erythematosus, and sarcoidosis.
30. The exposure to what irritant gases can cause ILD?
Chlorine, ammonia, and nitrogen dioxide.
31. What is idiopathic pulmonary fibrosis (IPF)?
A progressive inflammatory disease of an unknown cause that leads to varying degrees of fibrosis and honeycombing in severe cases.
32. What are two rare diseases that can lead to ILD?
(1) Lymphangioleiomyomatosis (LAM), and (2) Pulmonary Langerhans cell histiocytosis (PLCH).
33. Which two miscellaneous diffuse interstitial lung diseases involve recurrent pulmonary hemorrhage?
(1) Goodpasture’s syndrome, and (2) Idiopathic pulmonary hemosiderosis.
34. How is DLCO affected by interstitial lung disease?
IDL can result in an increase in DLCO.
35. What is expected during the physical examination of a patient with ILD?
An increased heart rate, blood pressure, and respiratory rate; cyanosis, digital clubbing, peripheral edema, venous distention, and a nonproductive cough.
36. What chest assessment findings can be expected with ILD?
Increased tactile and vocal fremitus, dull percussion note, bronchial breath sounds, crackles, pleural friction rub, and whispered pectoriloquy.
37. What could be found on a chest radiograph associated with ILD?
Bilateral reticulonodular pattern, irregularly shaped opacities, granulomas, cavity formation, honeycombing, and pleural effusion.
38. Why is the management of ILD so difficult?
Because there is often no specific cause.
39. What are the respiratory care treatment protocols for ILD?
Oxygen therapy, bronchopulmonary hygiene therapy, mechanical ventilation, pulmonary rehabilitation, research trials, lung transplantation, and palliative care.
40. What is idiopathic pulmonary fibrosis?
The formation of scar tissue in the connective tissue of the lungs without a known cause, resulting in severe chronic restrictive lung disease.
41. What is the definition of interstitial lung disease?
A group of respiratory disorders characterized by a dry, unproductive cough and dyspnea on exertion. The chest radiograph usually shows fibrotic infiltrates in the lower lobe.
42. What is occupational ILD?
Interstitial lung disease resulting from occupational exposure (e.g., asbestosis).
43. What is required to make a diagnosis of interstitial lung disease?
A lung biopsy is typically required to make a definitive diagnosis of interstitial lung disease.
44. What are the key diagnostic features of interstitial lung disease (ILD)?
The key diagnostic features of interstitial lung disease (ILD) include dyspnea at rest or with exertion; bilateral diffuse interstitial infiltrates on a chest radiograph, physiologic abnormalities of a restrictive lung defect, and histopathologic features of inflammation or fibrosis of the pulmonary parenchyma.
45. Which interstitial lung disease has an unknown cause?
Sarcoidosis
46. What is used to identify the type and etiology of interstitial lung disease?
Physicians perform many different tests to determine if a patient has interstitial lung disease (ILD). However, the patient’s medical history is most valuable in determining the specific type and etiology.
47. A physician is examining a patient whom she suspects has a form of interstitial lung disease (ILD). The patient is a smoker and presents with dyspnea, cough, fever, chills, and night sweats. The physical exam reveals digital clubbing. What disease might the patient have?
Patients with interstitial lung disease present with many different symptoms; however, the most likely diagnosis is idiopathic pulmonary fibrosis (IPF).
48. A physician is examining the chest radiograph of a patient she suspects has a form of interstitial lung disease (ILD). The radiograph shows eggshell calcification. What disease might the patient have?
The appearance of eggshell calcification on a chest radiograph is almost always unique to silicosis.
49. What are the indications for surgical lung biopsy in patients whom physicians suspect interstitial lung disease (ILD)?
Indications for surgical lung biopsy include a patient younger than 65 years; a history of fevers, weight loss, and sweats; a history of hemoptysis; symptoms and signs of peripheral vasculitis; a history of pneumothorax; a normal chest radiograph; and rapidly progressing disease.
50. What test can help physicians determine the needed biopsy sites for patients with suspected interstitial lung disease (ILD)?
Physicians use high-resolution computed tomography (HCRT) to help determine the best sites to biopsy in patients suspected of interstitial lung disease (ILD).
51. What is the primary objective of therapy in patients with interstitial lung disease (ILD)?
The most important tenet of therapy for patients with interstitial lung disease (ILD) is to remove the agent of injury, if possible. If the agent cannot be found and removed, suppressing inflammatory and cellular immune responses is the next priority.
52. What is the treatment for ILD dependent on?
The treatment method is dependent on the specific cause and symptoms.
53. Is coronavirus a potential cause of ILD?
Yes, ILD can occur secondary to coronavirus.
54. What is the most common drug administered for ILD?
Corticosteroids
55. Is lung scarring from ILD reversible?
No, once the damage has occurred, it is generally irreversible.
56. What symptom often limits exercise tolerance in patients with ILD?
Progressive dyspnea on exertion.
57. Why is ILD commonly classified as a restrictive lung disease?
Because lung compliance is reduced, leading to decreased lung volumes.
58. What happens to total lung capacity (TLC) in ILD?
Total lung capacity is decreased.
59. How does ILD affect lung compliance?
Lung compliance is reduced due to fibrosis and stiffening of lung tissue.
60. What diffusion abnormality is commonly seen in ILD?
Impaired diffusion of oxygen across the alveolar-capillary membrane.
61. Why is hypoxemia common in patients with ILD?
Thickening and fibrosis of the interstitium impair gas exchange.
62. When is hypoxemia most noticeable in early ILD?
During exercise or physical exertion.
63. What cardiovascular complication may develop in advanced ILD?
Pulmonary hypertension
64. How does ILD contribute to pulmonary hypertension?
Chronic hypoxemia causes pulmonary vasoconstriction and vascular remodeling.
65. What right-sided heart condition can result from advanced ILD?
Cor pulmonale
66. What digital finding is commonly associated with chronic ILD?
Digital clubbing
67. Why are fine crackles in ILD often described as “Velcro-like”?
They result from the sudden opening of stiff, fibrotic alveoli during inspiration.
68. What type of cough is most characteristic of ILD?
A dry, nonproductive cough.
69. Why are bronchodilators often less effective in ILD?
Because airflow obstruction is not the primary problem.
70. What imaging modality is most sensitive for detecting early ILD?
High-resolution computed tomography (HRCT)
71. What HRCT finding is characteristic of advanced fibrotic ILD?
Honeycombing
72. What does honeycombing represent pathologically?
Irreversible fibrosis with cystic airspaces.
73. Why is early diagnosis of ILD important?
Early intervention may slow disease progression before fibrosis becomes extensive.
74. What occupational exposure is associated with asbestosis?
Chronic inhalation of asbestos fibers.
75. What pleural abnormality is commonly associated with asbestosis?
Fibrocalcific pleural plaques
76. What environmental exposure commonly causes hypersensitivity pneumonitis?
Repeated inhalation of organic antigens.
77. Why is removing the offending antigen critical in hypersensitivity pneumonitis?
Continued exposure worsens inflammation and fibrosis.
78. What lab abnormality may be seen due to chronic hypoxemia in ILD?
Secondary polycythemia
79. Why do patients with ILD often develop tachypnea?
To compensate for reduced lung compliance and impaired gas exchange.
80. What breathing pattern may be observed in severe ILD?
Rapid, shallow breathing.
81. Why is pulmonary rehabilitation beneficial for ILD patients?
It improves exercise tolerance and quality of life.
82. What is the role of supplemental oxygen in ILD?
To treat hypoxemia and reduce cardiopulmonary strain.
83. Why is mechanical ventilation challenging in ILD?
Stiff lungs increase the risk of barotrauma and volutrauma.
84. What ventilatory strategy is often preferred in ILD?
Low tidal volume ventilation.
85. What is the long-term prognosis for most forms of progressive ILD?
Progressive decline in lung function with limited reversibility.
86. Why does ILD often lead to rapid oxygen desaturation during exertion?
Because the thickened interstitium limits oxygen diffusion during increased metabolic demand.
87. What happens to the alveolar-capillary membrane in ILD?
It becomes thickened and fibrotic, impairing gas exchange.
88. Why is fatigue a common complaint in patients with ILD?
Chronic hypoxemia and increased work of breathing increase energy expenditure.
89. What spirometry finding helps distinguish ILD from obstructive lung disease?
A reduced FVC with a normal or increased FEV₁/FVC ratio.
90. How does ILD affect residual volume (RV)?
Residual volume is usually normal or decreased.
91. Why is early ILD sometimes missed on a standard chest x-ray?
Radiographic changes may be subtle or absent in early disease.
92. What physical exam finding suggests advanced ILD with cardiac involvement?
Peripheral edema due to right-sided heart strain.
93. Why are infections particularly dangerous in patients with ILD?
Limited pulmonary reserve makes compensation difficult.
94. What role does smoking play in certain types of ILD?
It increases the risk and severity of fibrotic lung injury.
95. Why is patient history critical when evaluating suspected ILD?
Exposure history often identifies the underlying cause.
96. What ABG abnormality is most consistent with advanced ILD?
Chronic hypoxemia with respiratory alkalosis early, progressing to respiratory failure.
97. Why is cough suppression often ineffective in ILD?
The cough is driven by interstitial irritation rather than airway secretions.
98. What pulmonary complication may occur if ILD progresses unchecked?
Progressive respiratory failure.
99. Why is lung compliance a key concept in understanding ILD mechanics?
Reduced compliance explains increased work of breathing and dyspnea.
100. What is the ultimate limiting factor in end-stage ILD?
Severely impaired gas exchange due to extensive fibrosis.
Final Thoughts
Interstitial lung disease (ILD) represents a complex and diverse group of disorders that profoundly impact lung structure and function. By targeting the interstitium, these diseases reduce lung compliance, impair gas exchange, and often lead to progressive respiratory failure.
For respiratory therapists, ILD is a clinically significant condition that demands strong skills in assessment, imaging interpretation, pulmonary function analysis, and oxygen management.
A solid understanding of interstitial lung disease allows therapists to provide safer, more effective care, support long-term patient needs, and contribute meaningfully to the multidisciplinary management of chronic respiratory illness.
Written by:
John Landry is a registered respiratory therapist from Memphis, TN, and has a bachelor's degree in kinesiology. He enjoys using evidence-based research to help others breathe easier and live a healthier life.
References
- Miserocchi G. The pulmonary interstitium: an introductory review. Adv Exp Med Biol. 2003.
- Antoine MH, Mlika M. Interstitial Lung Disease. [Updated 2023 Jul 31]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025.