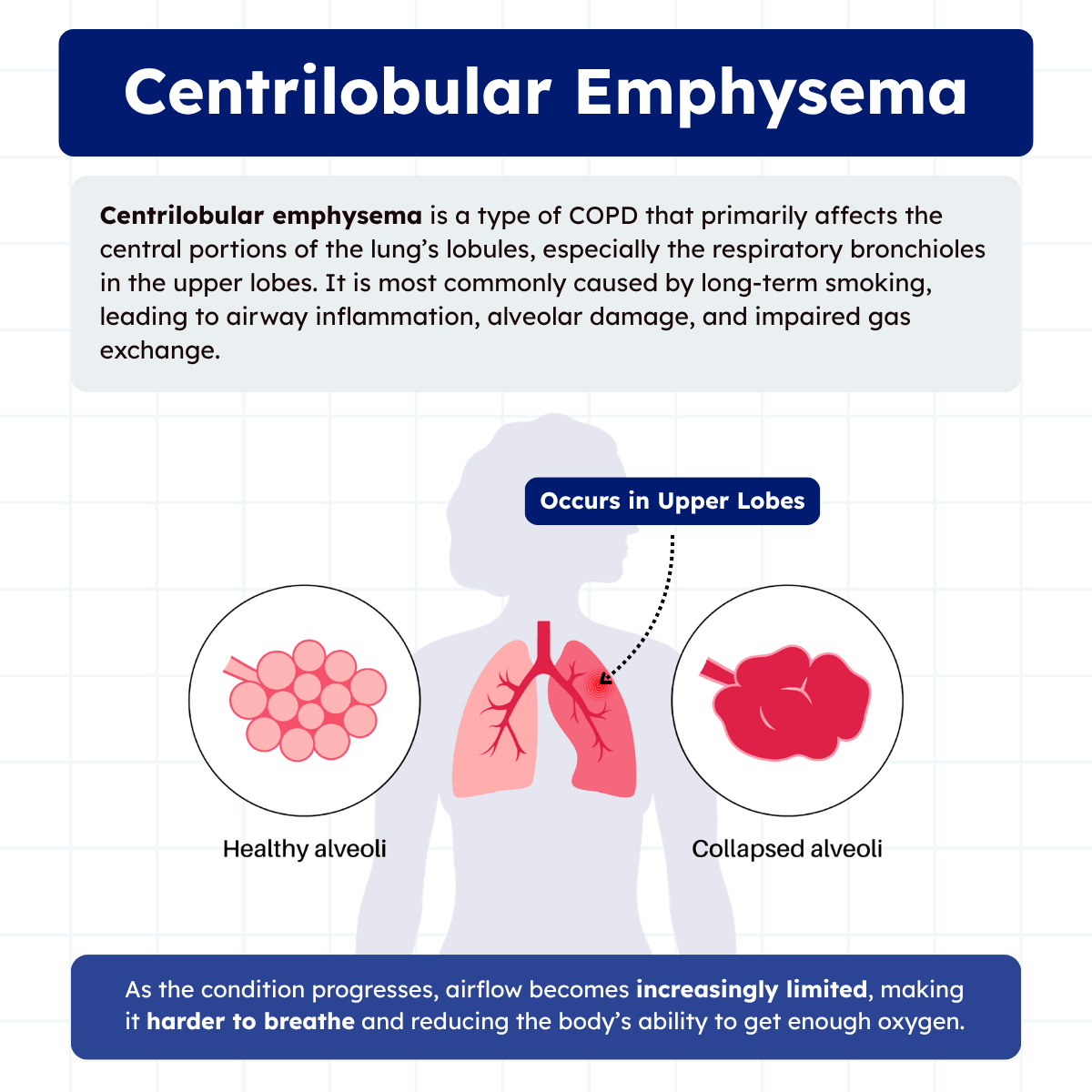
Centrilobular emphysema is a common form of chronic obstructive pulmonary disease (COPD) that primarily affects the central portions of the lung’s lobules, particularly in the upper lobes. It is most strongly associated with long-term cigarette smoking, which leads to inflammation, airway narrowing, and destruction of the surrounding alveolar walls.
As the disease progresses, these structural changes impair gas exchange and contribute to symptoms such as shortness of breath, chronic cough, and reduced exercise tolerance.
Understanding the underlying mechanisms and clinical features of centrilobular emphysema is essential for early recognition, proper management, and improved patient outcomes.
What Is Centrilobular Emphysema?
Centrilobular emphysema is a type of COPD that primarily affects the central portions of the lung’s secondary lobules, especially in the upper lobes. It is most commonly caused by long-term cigarette smoking, which leads to chronic inflammation and damage to the small airways known as bronchioles. Over time, this damage results in the destruction of nearby alveolar walls, reducing the surface area available for gas exchange.
Unlike other forms of emphysema, centrilobular emphysema tends to spare the distal alveoli in its early stages, with damage concentrated around the respiratory bronchioles.
As the condition progresses, airflow limitation worsens, leading to symptoms such as shortness of breath, chronic cough, and decreased exercise tolerance. Early detection and smoking cessation are key factors in slowing disease progression.
Distinguishing Features
The hallmark characteristic of centrilobular emphysema is its preferential involvement of the proximal respiratory bronchioles within each lobule. This contrasts with panlobular emphysema, which affects the entire acinus uniformly from the start.
On CT imaging, centrilobular emphysema appears as multiple small low-attenuation areas without visible walls. These areas do not extend to the pleural surface or interlobular septa. The disease shows a strong predilection for the upper lung zones, particularly the apical and posterior segments.
Pathologically, the respiratory bronchioles show dilation and destruction, while the distal alveoli remain relatively preserved in early stages. This creates a distinctive pattern that pathologists can identify on microscopic examination.
Prevalence and Epidemiology
Centrilobular emphysema accounts for approximately 95% of all emphysema cases related to smoking. It affects men more frequently than women, though this gap has narrowed as smoking rates among women have increased.
The condition typically develops in individuals over 50 years of age with a significant smoking history. Most patients have smoked at least 20 pack-years, though susceptibility varies based on genetic factors and other environmental exposures.
Note: Studies indicate that approximately 15-20% of chronic smokers develop clinically significant emphysema. The prevalence increases with age and cumulative smoke exposure.
Pathophysiology
Centrilobular emphysema develops through oxidative stress and inflammatory processes that selectively destroy respiratory bronchioles in the center of the secondary pulmonary lobule. The damage creates a characteristic pattern where destruction begins centrally and may extend outward while initially sparing distal alveoli and peripheral structures.
Alveolar Damage Mechanisms
Cigarette smoke and other inhaled toxins trigger an inflammatory cascade that releases proteolytic enzymes, particularly neutrophil elastase and matrix metalloproteinases. These enzymes break down elastin and collagen fibers in alveolar walls, which normally provide structural support and elastic recoil to lung tissue.
The body’s antiprotease defenses, especially alpha-1 antitrypsin, become overwhelmed by the excessive protease activity. This protease-antiprotease imbalance leads to progressive destruction of the extracellular matrix surrounding respiratory bronchioles.
Oxidative stress compounds the damage by inactivating remaining antiproteases and promoting further inflammatory cell recruitment. Chronic inflammation persists even after smoking cessation in many patients, perpetuating ongoing tissue destruction.
Centrilobular vs. Panlobular Emphysema
Centrilobular emphysema predominantly affects the proximal respiratory bronchioles in the center of the acinus, typically showing upper lobe predominance. The peripheral alveolar ducts and sacs remain relatively preserved in early disease stages.
Panlobular emphysema involves uniform destruction of the entire acinus from respiratory bronchiole to terminal alveoli. This pattern occurs most commonly in alpha-1 antitrypsin deficiency and demonstrates lower lobe predominance.
The anatomical distribution differs because centrilobular disease correlates strongly with cigarette smoke exposure, which has maximal impact on proximal airways. Panlobular emphysema results from genetic antiprotease deficiency affecting all alveolar structures equally.
Progression of Lung Injury
Initial damage creates small areas of destruction around individual respiratory bronchioles measuring 1-2 mm in diameter. These lesions gradually enlarge as proteolytic activity continues and adjacent damaged areas coalesce into larger bullae.
As emphysematous spaces expand, they compress surrounding healthier lung tissue and reduce functional alveolar surface area. The loss of elastic tissue decreases lung elastic recoil, causing air trapping and hyperinflation.
Advanced disease shows widespread destruction extending from centrilobular zones toward the periphery, though some distinction from panlobular patterns may persist. Pulmonary capillaries are destroyed along with alveolar walls, reducing the surface area available for gas exchange and contributing to ventilation-perfusion mismatch.
Causes and Risk Factors
Centrilobular emphysema develops primarily from cigarette smoking, though genetic predisposition and environmental exposures also contribute to disease risk and progression.
Smoking as a Major Risk Factor
Cigarette smoking accounts for approximately 85-90% of centrilobular emphysema cases. Tobacco smoke contains thousands of toxic chemicals that trigger chronic inflammation in the small airways and destroy alveolar walls. The inflammatory response generates oxidative stress and proteolytic enzymes that break down lung elastin faster than the body can repair it.
Pack-years serve as the standard measurement for smoking exposure, calculated by multiplying the number of cigarette packs smoked daily by years of smoking. Risk increases proportionally with pack-years, though individual susceptibility varies. Studies show that 15-20% of smokers develop clinically significant emphysema.
Note: Former smokers retain elevated risk compared to never-smokers, though quitting at any age slows disease progression. Secondhand smoke exposure also increases risk, particularly in childhood or with prolonged adult exposure.
Genetic Influences
Alpha-1 antitrypsin (AAT) deficiency represents the most significant genetic risk factor for early-onset emphysema. This inherited disorder reduces levels of AAT, a protective enzyme that inhibits neutrophil elastase from breaking down lung tissue. Individuals with severe AAT deficiency may develop emphysema in their 30s or 40s, even without smoking.
The condition follows an autosomal codominant pattern, with the most severe deficiency occurring in individuals with two Z alleles (PiZZ genotype). Carriers with one abnormal allele (PiMZ) show intermediate AAT levels and modestly increased emphysema risk when combined with smoking.
Note: Other genetic variations affect susceptibility to smoking-related lung damage, though their individual effects are smaller than AAT deficiency.
Environmental and Occupational Exposures
Biomass fuel smoke from cooking and heating increases emphysema risk in populations with limited access to clean energy sources. Coal, wood, and crop residue combustion releases particulate matter and toxic gases similar to tobacco smoke.
Occupational exposures to mineral dusts, chemical fumes, and vapors contribute to emphysema development. High-risk occupations include coal mining, hard rock mining, construction work, and manufacturing jobs involving cadmium, silica, or textile dust.
Note: Air pollution from traffic emissions and industrial sources shows associations with accelerated lung function decline and increased emphysema prevalence in long-term exposure studies.
Clinical Presentation
Centrilobular emphysema typically manifests in current or former smokers during their fifth or sixth decade of life. The disease presents with progressive respiratory symptoms that worsen over time, along with characteristic physical examination findings.
Common Symptoms
Patients with centrilobular emphysema most commonly report progressive dyspnea on exertion as their initial complaint. This breathlessness typically develops gradually over years and eventually interferes with daily activities.
Chronic productive cough is frequently present, particularly in the morning hours. The sputum is usually mucoid but may become purulent during acute exacerbations. Many patients describe increased sputum production during winter months.
Wheezing occurs in approximately 30-40% of patients and reflects airway obstruction. Some individuals experience chest tightness or a sensation of incomplete lung expansion. Weight loss may develop in advanced cases due to increased work of breathing and decreased appetite.
Physical Examination Findings
Early-stage centrilobular emphysema may show minimal physical examination abnormalities. As the disease progresses, patients develop a barrel-shaped chest with increased anteroposterior diameter.
Decreased breath sounds are evident on auscultation, particularly in the upper lung fields where centrilobular emphysema predominantly affects tissue. Prolonged expiratory phase is a hallmark finding that reflects airflow limitation. Wheezes or rhonchi may be audible during normal breathing or forced expiration.
Patients often exhibit pursed-lip breathing to maintain positive airway pressure during expiration. Use of accessory respiratory muscles becomes apparent in moderate to severe disease. Hyperresonance to percussion reflects lung hyperinflation.
Note: Clubbing is not typically associated with uncomplicated centrilobular emphysema. Cyanosis appears only in advanced disease with significant hypoxemia.
Complications
Acute exacerbations represent the most frequent complication, characterized by worsening dyspnea, increased sputum production, and sputum purulence. These episodes often require medical intervention and may lead to respiratory failure.
Pneumothorax occurs more frequently in emphysema patients than in the general population due to rupture of bullae. Patients present with sudden-onset chest pain and worsening breathlessness.
Chronic respiratory failure develops as the disease progresses, manifesting as hypoxemia and potentially hypercapnia. Cor pulmonale results from pulmonary hypertension secondary to chronic hypoxemia and loss of pulmonary capillary bed. This condition presents with peripheral edema, elevated jugular venous pressure, and right ventricular heave.
Note: Recurrent respiratory infections are common due to impaired mucociliary clearance and retained secretions. Polycythemia may develop as a compensatory response to chronic hypoxemia.
Diagnostic Methods
Centrilobular emphysema requires a combination of imaging studies and functional assessments to establish an accurate diagnosis. High-resolution computed tomography serves as the primary imaging modality, while pulmonary function tests quantify the extent of airflow limitation.
Imaging Techniques
High-resolution computed tomography (HRCT) is the gold standard for diagnosing centrilobular emphysema. HRCT reveals areas of low attenuation in the central portions of secondary pulmonary lobules, predominantly affecting the upper lobes. The characteristic pattern shows destruction of respiratory bronchioles while sparing the peripheral alveolar ducts and sacs.
Standard chest radiographs may show hyperinflation and flattened diaphragms in advanced cases. However, chest X-rays lack sensitivity for detecting early-stage disease. CT scans can detect emphysema before patients develop significant symptoms or spirometric abnormalities.
Quantitative CT analysis measures the extent of emphysematous destruction using density mask techniques. Radiologists typically define emphysema as areas with attenuation values below -950 Hounsfield units. This objective measurement helps clinicians monitor disease progression over time.
Pulmonary Function Testing
Spirometry demonstrates irreversible airflow obstruction with reduced FEV1/FVC ratio (typically below 0.70). The FEV1 measurement indicates the severity of obstruction and helps guide treatment decisions. Post-bronchodilator testing shows minimal improvement, distinguishing emphysema from reversible conditions.
Lung volume measurements reveal air trapping with increased residual volume and total lung capacity. The diffusing capacity for carbon monoxide (DLCO) is characteristically reduced due to alveolar-capillary membrane destruction. DLCO measurements often correlate with the anatomic extent of emphysema seen on CT imaging.
Differential Diagnosis
Clinicians must distinguish centrilobular emphysema from panlobular emphysema, which shows uniform destruction of the entire acinus and predominates in the lower lobes. Alpha-1 antitrypsin deficiency should be considered in patients with basilar-predominant disease or those diagnosed before age 45.
Chronic bronchitis may coexist with centrilobular emphysema, but represents a distinct clinical entity defined by chronic productive cough. Bronchiectasis can mimic emphysema on imaging but shows bronchial wall thickening and mucus plugging rather than parenchymal destruction.
Treatment Options
Managing centrilobular emphysema requires a combination of behavioral changes, pharmaceutical interventions, and structured exercise programs to slow disease progression and improve quality of life.
Lifestyle Modifications
Smoking cessation is the most critical intervention for patients with centrilobular emphysema. Continued tobacco use accelerates lung tissue destruction and worsens symptoms. Patients who quit smoking experience slower disease progression and improved respiratory function compared to those who continue.
Healthcare providers should connect patients with smoking cessation programs that include counseling and nicotine replacement therapy. Support groups and behavioral interventions increase success rates.
Avoiding environmental irritants is equally important. Patients should minimize exposure to secondhand smoke, air pollution, chemical fumes, and dust. Using air purifiers at home and wearing masks in polluted areas can reduce respiratory irritation.
Maintaining a healthy weight supports respiratory function. Underweight patients may need nutritional counseling to prevent muscle wasting, while overweight patients benefit from weight management to reduce breathing effort.
Medications
Bronchodilators are the primary medications for centrilobular emphysema. Short-acting bronchodilators provide quick relief during acute symptoms, while long-acting versions offer sustained symptom control throughout the day.
Inhaled corticosteroids may be prescribed for patients with frequent exacerbations or evidence of airway inflammation. These medications reduce inflammation in the airways and can decrease the frequency of symptom flare-ups when combined with bronchodilators.
Phosphodiesterase-4 inhibitors help reduce inflammation and may be added for patients with severe disease and chronic bronchitis. Antibiotics and oral corticosteroids are reserved for treating acute exacerbations.
Note: Patients with low blood oxygen levels may require supplemental oxygen therapy. Oxygen use improves survival rates and exercise capacity in individuals with significant hypoxemia.
Pulmonary Rehabilitation
Pulmonary rehabilitation is a comprehensive program that combines exercise training, education, and behavioral modification. These programs typically last 6 to 12 weeks and involve supervised sessions two to three times per week.
Exercise training focuses on aerobic activities like walking or cycling along with strength training for upper and lower body muscles. Patients learn breathing techniques such as pursed-lip breathing and diaphragmatic breathing to improve ventilation efficiency.
Education components cover disease management, proper medication use, and recognizing early signs of exacerbations. Nutritional counseling and psychological support address the broader impacts of chronic lung disease.
Studies show pulmonary rehabilitation reduces shortness of breath, increases exercise tolerance, and decreases hospital admissions. Benefits persist for months after program completion, though maintenance exercises are recommended.
Disease Management and Prognosis
Effective management requires consistent medical oversight and lifestyle modifications, while prognosis depends on disease severity, smoking cessation, and comorbid conditions.
Long-Term Monitoring
Patients with centrilobular emphysema require regular pulmonary function testing every 6 to 12 months to track disease progression. Spirometry measurements help clinicians assess FEV1 decline rates and adjust treatment protocols accordingly.
Chest imaging through CT scans should be performed every 2 to 3 years in stable patients, or more frequently if symptoms worsen. These scans detect bullae formation, assess disease distribution, and identify complications such as pneumothorax or lung cancer.
Oxygen saturation monitoring becomes critical as disease advances. Patients should undergo arterial blood gas analysis when resting oxygen saturation falls below 92% or during exercise desaturation events. Healthcare providers also monitor for signs of pulmonary hypertension and cor pulmonale through echocardiography when indicated.
Factors Affecting Prognosis
Smoking cessation represents the most significant modifiable factor influencing survival rates. Patients who quit smoking demonstrate slower FEV1 decline compared to those who continue, with studies showing an average difference of 20 to 30 mL per year.
Baseline lung function at diagnosis strongly predicts outcomes. Patients with FEV1 values above 50% predicted have 5-year survival rates exceeding 80%, while those with FEV1 below 30% face significantly reduced survival without intervention.
Age at diagnosis, body mass index, and exercise capacity measured by 6-minute walk distance provide additional prognostic information. Younger patients with preserved nutritional status and maintained physical activity levels generally experience better long-term outcomes. The presence of frequent exacerbations, defined as two or more per year, correlates with accelerated disease progression and increased mortality risk.
Management of Severe Cases
Severe centrilobular emphysema requires intensive interventions including long-term oxygen therapy for patients with resting PaO2 below 55 mmHg. Oxygen therapy improves survival and reduces hospitalization frequency when used at least 15 hours daily.
Lung volume reduction surgery benefits selected patients with upper lobe predominant disease and low exercise capacity. This procedure removes damaged tissue to improve respiratory mechanics and exercise tolerance. Bronchoscopic valve placement offers a less invasive alternative for appropriate candidates with heterogeneous disease distribution.
Lung transplantation remains an option for end-stage disease in patients under 65 years old without significant comorbidities. Referral to transplant centers should occur when FEV1 falls below 25% predicted or when patients develop secondary pulmonary hypertension. Palliative care consultation helps manage symptoms and quality of life issues in patients with advanced disease not eligible for surgical interventions.
Complications and Comorbidities
Centrilobular emphysema leads to progressive lung damage that increases susceptibility to pulmonary hypertension and recurrent respiratory infections. These complications often accelerate disease progression and significantly impact patient outcomes.
Pulmonary Hypertension
Destruction of capillary beds in the centrilobular regions reduces the total cross-sectional area of the pulmonary vasculature. This structural loss forces blood through fewer vessels, elevating resistance and pressure within the pulmonary circulation.
Chronic hypoxemia from impaired gas exchange triggers vasoconstriction in pulmonary arteries. The body attempts to redirect blood flow to better-ventilated lung areas, but sustained vasoconstriction becomes pathologic. Over time, arterial walls thicken and remodel, making the elevated pressure permanent.
Right ventricular strain develops as the heart works harder to pump blood through high-resistance pulmonary vessels. Patients may experience worsening dyspnea, fatigue, and peripheral edema as right heart function deteriorates. Early detection through echocardiography or right heart catheterization allows for targeted therapies including oxygen supplementation and pulmonary vasodilators.
Respiratory Infections
Damaged airways and impaired mucus clearance create ideal conditions for bacterial colonization. Patients with centrilobular emphysema experience more frequent and severe lower respiratory tract infections than healthy individuals.
Common pathogens include Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis. Viral infections, particularly influenza and respiratory syncytial virus, often precipitate acute exacerbations that require hospitalization.
Reduced immune surveillance in destroyed lung tissue allows infections to establish quickly. Inflammation from infection further narrows already compromised airways and increases mucus production. Each infection episode can cause additional permanent lung damage, accelerating the decline in respiratory function.
Note: Vaccination against influenza and pneumococcal disease reduces infection frequency. Prompt antibiotic treatment during exacerbations prevents progression to respiratory failure.
Current Research and Future Directions
Researchers are investigating novel biomarkers to detect centrilobular emphysema earlier and monitor disease progression more accurately. Advanced imaging techniques, including photon-counting CT and hyperpolarized gas MRI, show promise in identifying structural changes before symptoms appear.
Gene therapy approaches targeting alpha-1 antitrypsin deficiency are in clinical trials. These studies aim to restore normal protein levels in patients with genetic predispositions to emphysema. Several pharmaceutical companies are developing augmentation therapies that could slow or halt disease progression.
Key Research Areas:
- Stem cell therapy to regenerate damaged lung tissue
- Anti-inflammatory agents targeting specific cytokines
- Protease inhibitor medications beyond current treatments
- Personalized medicine based on genetic profiles
Scientists are exploring the role of the lung microbiome in disease development. Studies suggest that bacterial colonization patterns may influence inflammation and tissue destruction rates. This research could lead to probiotic or antimicrobial interventions.
Artificial intelligence algorithms are being developed to analyze CT scans and predict disease trajectories. Machine learning models can identify subtle patterns that human observers might miss, potentially enabling more precise treatment planning.
Clinical trials are testing combination therapies that address multiple disease mechanisms simultaneously. Researchers are evaluating drugs that reduce oxidative stress while enhancing tissue repair processes.
The development of less invasive lung volume reduction techniques continues to advance. Bronchoscopic approaches using valves, coils, and vapor ablation offer alternatives to traditional surgery for selected patients. Long-term outcome data from these interventions will help refine patient selection criteria.
FAQs About Centrilobular Emphysema
What Is the Difference Between Emphysema and Centrilobular Emphysema?
Emphysema is a broad term for a type of COPD characterized by destruction of the alveoli, leading to impaired gas exchange. Centrilobular emphysema is a specific subtype that primarily affects the central portions of the lung lobules, particularly the respiratory bronchioles in the upper lobes.
While emphysema can include different patterns of lung damage, centrilobular emphysema is most commonly associated with smoking and has a distinct distribution within the lungs compared to other forms.
What Are the Risk Factors for Centrilobular Emphysema?
The primary risk factor for centrilobular emphysema is long-term cigarette smoking, which causes chronic inflammation and damage to the small airways. Other risk factors include exposure to air pollution, occupational dust and chemicals, and secondhand smoke.
Age is also a factor, as lung damage accumulates over time. A history of chronic bronchitis or repeated respiratory infections may increase risk, as these conditions contribute to ongoing airway irritation and structural damage within the lungs.
What Is the Life Expectancy of a Person with Centrilobular Emphysema?
Life expectancy varies depending on the severity of the disease, overall health, and whether the person continues to smoke. Individuals who quit smoking and follow appropriate treatment plans may live for many years with manageable symptoms.
In contrast, advanced disease with severe airflow limitation and frequent exacerbations can shorten life expectancy. Early diagnosis, proper medical care, and lifestyle modifications such as smoking cessation and pulmonary rehabilitation play a key role in improving long-term outcomes.
What Is Mild Centrilobular Emphysema?
Mild centrilobular emphysema refers to the early stage of the disease, where structural damage to the lungs is limited and symptoms may be minimal or absent. It is often detected incidentally on imaging studies such as a CT scan.
At this stage, airflow limitation is usually mild, and lung function may still be relatively preserved. Early intervention, especially smoking cessation, is critical because it can significantly slow disease progression and help maintain lung function over time.
How Serious Is Centrilobular Emphysema?
Centrilobular emphysema can range from mild to severe, but it is considered a serious and progressive lung condition. As the disease advances, the destruction of lung tissue leads to worsening airflow limitation and reduced oxygen exchange. This can result in significant shortness of breath, decreased physical activity, and reduced quality of life.
In severe cases, complications such as respiratory failure may occur. The seriousness largely depends on disease stage, smoking status, and how well it is managed.
Can Covid-19 Cause Centrilobular Emphysema?
COVID-19 does not directly cause centrilobular emphysema, but severe infection can lead to lung damage that may worsen existing respiratory conditions. In some cases, COVID-19 can cause inflammation and scarring in the lungs, which may mimic or contribute to emphysema-like changes.
However, centrilobular emphysema is most strongly associated with long-term smoking rather than viral infections. Patients with preexisting emphysema may experience more severe outcomes if they contract COVID-19.
Is Centrilobular Emphysema a Type of Cancer?
Centrilobular emphysema is not a type of cancer. It is a chronic lung disease that involves the destruction of airspaces within the lungs, leading to impaired breathing and gas exchange.
However, smoking is a major risk factor for both emphysema and lung cancer, so individuals with centrilobular emphysema may have an increased risk of developing lung cancer.
Note: Regular medical follow-up and screening may be recommended, especially for those with a significant smoking history.
How Fast Does Centrilobular Emphysema Progress?
The progression of centrilobular emphysema varies widely from person to person. It often develops slowly over many years, especially in individuals who continue to smoke. The rate of progression depends on factors such as smoking status, environmental exposures, and overall health.
Quitting smoking can significantly slow the progression of the disease. Without intervention, lung function may decline steadily, leading to worsening symptoms and increased risk of complications over time.
Is Centrilobular Emphysema the Same as COPD?
Centrilobular emphysema is not the same as COPD, but it is a type of COPD. COPD is an umbrella term that includes chronic bronchitis and emphysema, both of which cause airflow limitation.
Centrilobular emphysema is one specific form of emphysema characterized by damage to the central parts of the lung lobules. While all patients with centrilobular emphysema have COPD, not all patients with COPD have this particular subtype of emphysema.
Is Centrilobular Emphysema Reversible?
Centrilobular emphysema is not reversible because the damage to the alveoli is permanent. Once lung tissue is destroyed, it cannot regenerate. However, the progression of the disease can be slowed with appropriate management.
Smoking cessation is the most important step, along with medications, pulmonary rehabilitation, and lifestyle changes. These interventions can help improve symptoms, enhance quality of life, and reduce the risk of complications, even though the underlying damage cannot be undone.
Final Thoughts
Centrilobular emphysema is a progressive lung condition that can significantly impact a person’s ability to breathe and carry out daily activities. Because it is most often linked to long-term smoking, prevention and early intervention play a critical role in limiting its severity.
Recognizing the signs and understanding how the disease affects the lungs can help guide timely diagnosis and appropriate treatment. While the damage caused by emphysema cannot be reversed, lifestyle changes, medical therapy, and supportive care can improve symptoms and enhance quality of life.
Written by:
John Landry is a registered respiratory therapist from Memphis, TN, and has a bachelor's degree in kinesiology. He enjoys using evidence-based research to help others breathe easier and live a healthier life.
References
- Anderson AE Jr, Foraker AG. Centrilobular emphysema and panlobular emphysema: two different diseases. Thorax. 1973.

